

Journal of Water, Sanitation, and Hygiene for Development 2020 10 (2): 298–308
Household stored water quality in an intermittent water supply network in Panama
Carlos Gonzalez, John Erickson, Karina Chavarría, Kara Nelson, Amador Goodridge.
Abstract:
Safe water storage is critical to preserve water quality, especially when intermittent piped drinking water supply creates a need for household storage. This study characterized household stor-age practices and stored water quality in 94 households (N = 94) among four peri-urban neighborhoods in Arraiján, Panama with varying degrees of supply intermittency. We found that 18 (19.1%) households stored drinking water in unsafe containers. Forty-four (47%) samples of household stored drinking water had residual chlorine levels <0.2 mg/L. While 33 (35.1%) samples were positive for total coliform bacteria, only 23 (24.4%) had >10 most probable number (MPN)/100 mL total coliform bacteria. Eight (44%) samples were positive for Escherichia coli, whereas only one (1.3%) sample from the safe containers was positive. Twenty-nine (30.9%) samples had >500 MPN/mL heterotrophic plate count bacteria. These findings suggest that longer supply interruptions were associated with longer storage times and lower chlorine residual, which were associated with higher concentrations of indi-cator bacteria. This is one of the first studies in the Central-Amer-ican region to show an association between the lack of turnover (replacement with fresh water) and greater contamination during household water storage. Thus, when drinking water supply is not completely continuous and household storage is required, decreasing the time between supply periods can facilitate safer water storage. Public awareness and education are also recom-mended to increase hygiene practices during water collection and storage.
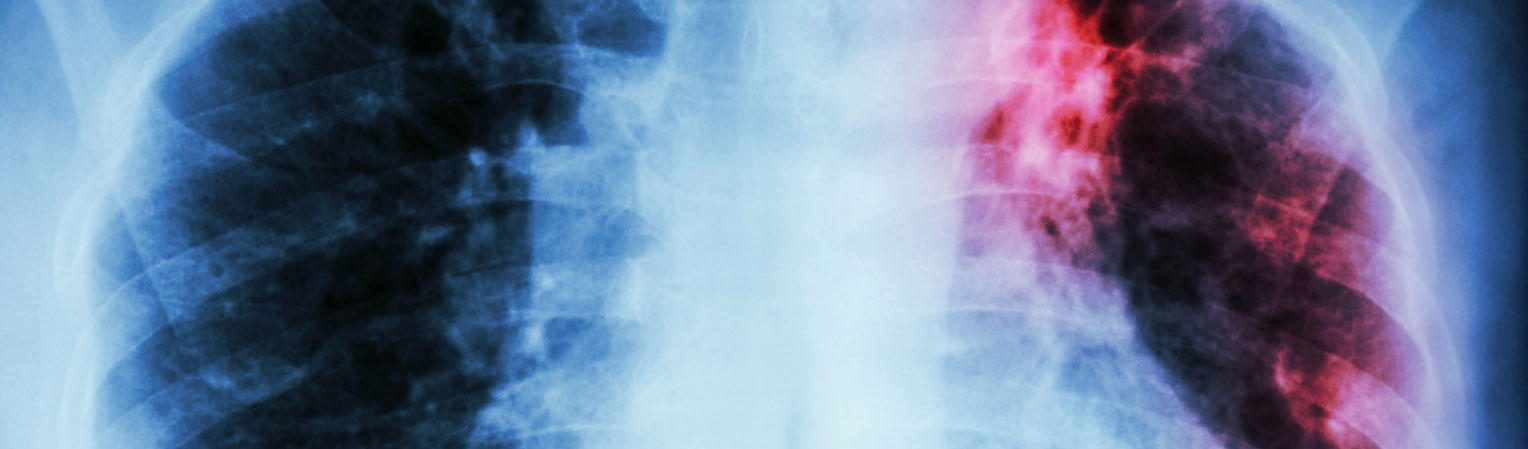
BMC Medicine (2018) 16:196
Global variation in bacterial strains that cause tuberculosis disease: a systematic review and meta-analysis
Kirsten E Wiens, Lauren P Woyczynski, Jorge R Ledesma, Jennifer M Ross, Roberto Zenteno-Cuevas, Amador Goodridge, Irfan Ullah,
Barun Mathema, Joel Fleury Djoba Siawaya, Molly H Biehl, Sarah E Ray, Natalia V Bhattacharjee, Nathaniel J Henry, Robert C Reiner Jr,
Hmwe H Kyu, Christopher J L Murray, Simon I Hay
Abstract:
Background: The host, microbial, and environmental factors that contribute to variation in tuberculosis (TB) disease are incompletely understood. Accumula-ting evidence suggests that one driver of geographic variation in TB disease
is the local ecology of mycobacterial genotypes or strains, and there is a need for a comprehensive and systematic synthesis of these data. The objectives of this study were to (1) map the global distribution of genotypes that cause
TB disease and (2) examine whether any epidemiologically relevant clinical characteristics were associated with those genotypes.
Methods: We performed a systematic review of PubMed and Scopus to create a comprehensive dataset of human TB molecular epidemiology studies that used representative sampling techniques. The methods were developed
according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). We extracted and synthesized data from studies that reported prevalence of bacterial genotypes and from studies that reported clinical characteristics associated with those genotypes.
Results: The results of this study are twofold. First, we identified 206 studies for inclusion in the study, representing over 200,000 bacterial isolates collected over 27 years in 85 countries. We mapped the genotypes and found that,
consistent with previously published maps, Euro-American lineage 4 and East Asian lineage 2 strains are widespread, and West African lineages 5 and 6 strains are geographically restricted. Second, 30 studies also reported transmission chains and 4 reported treatment failure associated with genotypes. We performed a meta-analysis and found substantial heterogeneity across studies. However, based on the data available, we found that lineage 2 strains may be associated with increased risk of transmission chains, while lineages 5 and 6 strains may be associated with reduced risk, compared with lineage 4 strains.
Conclusions: This study provides the most comprehensive systematic analysis of the evidence for diversity in bacterial strains that cause TB disease. The results show both geographic and epidemiological differences between strains, which could inform our understanding of the global burden of TB. Our findings also highlight the challenges of collecting the clinical data required to inform TB diagnosis and treatment. We urge future national TB programs and research efforts to prioritize and reinforce clinical data collection in study designs and results dissemination.
Emerging Infectious Diseases -2019;25(3):507-514.
Simplified Model to Survey Tuberculosis Transmission in
Countries without Systematic Molecular Epidemiology Programs
Juan Domínguez, Fermín Acosta, Laura Pérez-Lago, Dilcia Sambrano, Victoria Batista, Carolina De La Guardia, Estefanía Abascal,
Álvaro Chiner-Oms, Iñaki Comas, Prudencio González, Jaime Bravo, Pedro Del Cid, Samantha Rosas, Patricia Muñoz, Amador Goodridge,
Darío García de Viedma.
Abstract:
Systematic molecular/genomic epidemiology studies for tuberculosis surveillance cannot be implemented in many countries. We selected Panama as a model for an alternative strategy. Mycobacterial interspersed repetitive unit–variable-number tandem-repeat (MIRU-VNTR) analysis revealed a high proportion (50%) of Mycobacterium tuberculosis isolates included in 6 clusters (A–F) in 2 provinces (Panama and Colon). Cluster A corresponded to the Beijing sublineage. Whole-genome sequencing (WGS) differentiated clusters due to active recent transmission, with low single-nucleotide polymorphism–based diversity (cluster C), from clusters involving long-term prevalent strains with higher diversity (clusters A, B). Prospective application in Panama of 3 tailored strain–specific PCRs targeting marker single-nucleotide polymorphisms identified from WGS data revealed that 31.4% of incident cases involved strains A–C and that the Beijing strain was highly represented and restricted mainly to Colon. Rational integration of MIRU-VNTR, WGS, and tailored strain–specific PCRs could be a new model for tuberculosis surveillance in countries without molecular/genomic epidemiology programs.

Tel: (507) 5170700 - Fax: (507) 5070020 - EFax: (507) 5170701 | INDICASAT - AIP | Edificio 219, Ciudad del Saber | Clayton, Apartado 0843-01103 | Panamá 5 Panamá, Rep. de Panamá.
© Copyright 2014. INDICASAT AIP. Todos los derechos reservados.
INDICASAT.org.pa

Quienes somos
Síguenos